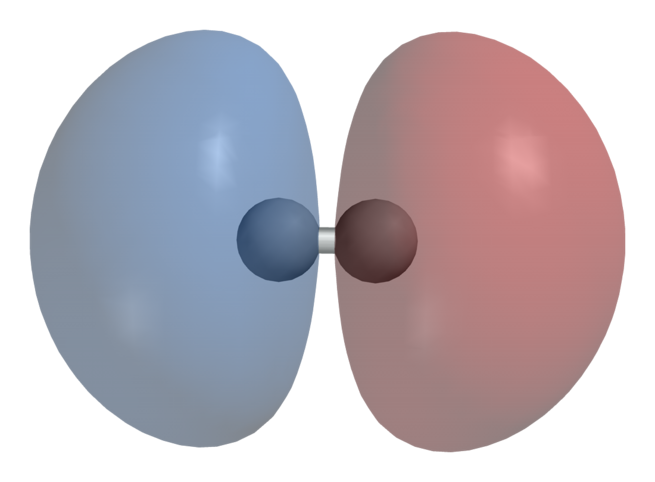
Main Difference
The main difference between Sigma Bond and Pi Bond is that the Sigma Bond is a strongest type of covalent chemical bond; formed by head-on overlapping between atomic orbitals. Sigma bonding for diatomic molecules (using the language and tools of symmetry groups):σ-bond is symmetrical with respect to rotation about the bond axis and Pi Bond is a Type of covalent chemical bond
-
Sigma Bond
In chemistry, sigma bonds (σ bonds) are the strongest type of covalent chemical bond. They are formed by head-on overlapping between atomic orbitals. Sigma bonding is most simply defined for diatomic molecules using the language and tools of symmetry groups. In this formal approach, a σ-bond is symmetrical with respect to rotation about the bond axis. By this definition, common forms of sigma bonds are s+s, pz+pz, s+pz and dz2+dz2 (where z is defined as the axis of the bond or the internuclear axis).
Quantum theory also indicates that molecular orbitals (MO) of identical symmetry actually mix or hybridize. As a practical consequence of this mixing of diatomic molecules, the wavefunctions s+s and pz+pz molecular orbitals become blended. The extent of this mixing (or hybridization or blending) depends on the relative energies of the MOs of like symmetry.
For homodiatomics(homonuclear diatomic molecules), bonding σ orbitals have no nodal planes at which the wavefunction is zero, either between the bonded atoms or passing through the bonded atoms. The corresponding antibonding, or σ* orbital, is defined by the presence of one nodal plane between the two bonded atoms.
Sigma bonds are the strongest type of covalent bonds due to the direct overlap of orbitals, and the electrons in these bonds are sometimes referred to as sigma electrons.The symbol σ is the Greek letter sigma. When viewed down the bond axis, a σ MO has a circular symmetry, hence resembling a similarly sounding “s” atomic orbital.
Typically, a single bond is a sigma bond while a multiple bond is composed of one sigma bond together with pi or other bonds. A double bond has one sigma plus one pi bond, and a triple bond has one sigma plus two pi bonds.
-
Pi Bond
Pi bonds (π bonds) are covalent chemical bonds where two lobes of an orbital on one atom overlap two lobes of an orbital on another atom and this overlap occurs laterally. Each of these atomic orbitals has zero electron density at a shared nodal plane, passing through the two bonded nuclei. The same plane is also a nodal plane for the molecular orbital of the pi bond. Pi Bonds can form in double and triple bonds but do not form in single bonds in most cases.
The Greek letter π in their name refers to p orbitals, since the orbital symmetry of the pi bond is the same as that of the p orbital when seen down the bond axis. One common form of this sort of bonding involves p orbitals themselves, though d orbitals also engage in pi bonding. This latter mode forms part of the basis for metal-metal multiple bonding.
Pi bonds are usually weaker than sigma bonds. The C-C double bond, composed of one sigma and one pi bond, has a bond energy less than twice that of a C-C single bond, indicating that the stability added by the pi bond is less than the stability of a sigma bond. From the perspective of quantum mechanics, this bond’s weakness is explained by significantly less overlap between the component p-orbitals due to their parallel orientation. This is contrasted by sigma bonds which form bonding orbitals directly between the nuclei of the bonding atoms, resulting in greater overlap and a strong sigma bond.
Pi bonds result from overlap of atomic orbitals that are in contact through two areas of overlap. Pi bonds are more diffuse bonds than the sigma bonds. Electrons in pi bonds are sometimes referred to as pi electrons. Molecular fragments joined by a pi bond cannot rotate about that bond without breaking the pi bond, because rotation involves destroying the parallel orientation of the constituent p orbitals.
For homonuclear diatomic molecules, bonding π molecular orbitals have only the one nodal plane passing through the bonded atoms, and no nodal planes between the bonded atoms. The corresponding antibonding, or π* (“pi-star”) molecular orbital, is defined by the presence of an additional nodal plane between these two bonded atoms.
