Main Difference
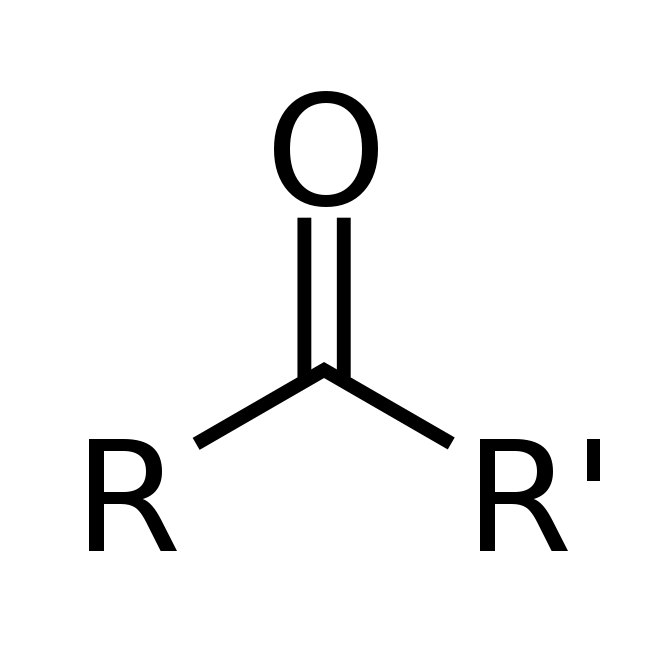
The main difference between Ketone and Phenone is that the Ketone is a class of organic compounds having structure RCOR´ and Phenone is a Wiktionary redirect.
-
Ketone
In chemistry, a ketone (alkanone) is an organic compound with the structure RC(=O)R’, where R and R’ can be a variety of carbon-containing substituents. Ketones and aldehydes are simple compounds that contain a carbonyl group (a carbon-oxygen double bond). They are considered “simple” because they do not have reactive groups like −OH or −Cl attached directly to the carbon atom in the carbonyl group, as in carboxylic acids containing −COOH. Many ketones are known and many are of great importance in industry and in biology. Examples include many sugars (ketoses) and the industrial solvent acetone, which is the smallest ketone.
-
Ketone (noun)
A homologous series of organic molecules whose functional group is an oxygen atom joined to a carbon atom—by a double bond—in a carbon-hydrogen based molecule.
-
Phenone (noun)
Any aromatic ketone containing a phenyl group directly attached to the carbonyl group